The purpose of Sterile Barrier Testing
The sterile barrier is the external barrier (some examples can be Tyvek material or Polyurethane seal) which separates the internal components, powders, medicines etc from the elements. When we say barrier, it is normally a vacuum sealed package or polyurethane sealing which protects the internal to the external.
In most cases, it is either water resistant or water proof.
Hence it is crucial that the sterile barrier is never compromised.
A key function of a sterile barrier system is to maintain the safety of a terminally sterilised medical device until its point of use - typically in a healthcare setting.
QRA International's method of Sterile Barrier Testing
QRA International's network of Laboratories can evaluate the sterile integrity of your final packaging design through a series of seal integrity and seal strength testing outlined in ISO 11607 - Packaging for Terminally Sterilised Medical Devices.
Seal integrity testing identifies any leaks around the seal area of your packaging system.Seal integrity testing identifies any leaks around the seal area of your packaging system. Seal integrity testing is followed by seal strength testing to evaluate the mechanical strength of your packaging system and the force needed to separate and open the seal. A high numerical force value could indicate challenges in opening your packaging system by hand, and a low numerical force value could indicate poor bonding of materials.
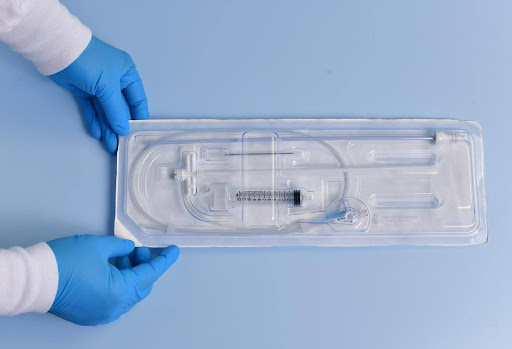
Sterile Barrier of a Sealed Catheter
Tyvek Seal and Polyurethane Packaging
form the Sterile Barrier System
QRA International Medical Device Testing
Helps you to
Validate your final packaging system<
Ensure proper device performance during the manufacturing process.
Establish material compatability and proper bonding to maintain the sterile environment throughout the shelf life of your product.
Ensure the seal of your packaging system does not change due to environmental stressors such as distribution and shelf life.
