IQ OQ and PQ Services to Validate your Test Equipment
QRA is the preferred laboratory services partner for many companies in the biomedical, medical devices and pharmaceutical industries in Singapore and internationally.
Our customers come from Japan, Singapore, Australia and USA.
Our 2 Laboratories comprising close 20 test chambers are running 24 hours a
day,7 days a week to provide accelerated ageing with controlled temperature and
humidity according to major international standards.
Before starting each test, QRA provides a comprehensive Calibration Report for each
chamber and also for the test sensors (temperature and humidity) with full ISO 17025
traceability. The Calibration Report of the chamber is done every year and the calibration report of the sensors and recording instrumentation every 2 years.
Upon start, interim durations (monthly) and completion of these tests,
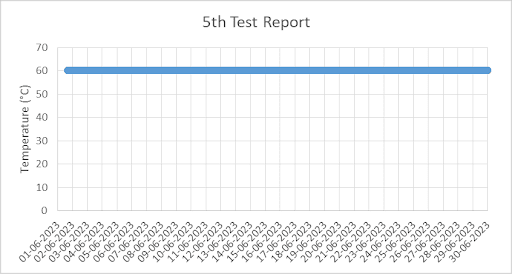
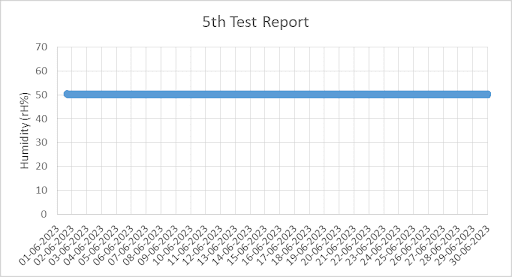
QRA provides detailed test reports with pictures and graphs to our clients.

Loading of Test Chamber with client samples
Sample taken from Test Report
4. Graph for Temperature and Humidity from 1st Jun 2023 to 30th Jun 2023 (taken from Chart Recorder)
Accelerated Ageing in Biomedical and Pharmaceutical Testing
Accelerated ageing tests are commonly used to verify that a new product can be stored
for an extended period of time (2, 3 5 or 8 years) in the shelf, storage facility or clinic without major loss in its
- function
- form
- efficacy
- texture
for the lifespan duration of the product (up to 8 years).
To accelerate the effects of time, the products are subjected to higher temperatures and
humidity conditions that it would normally encounter during its shipping, storage and
operational life cycle.
The objective of accelerated ageing is to provide assurance that the product will meet its expected lifetime and reliability goals in as short as possible a time and tweak or
redesign the product at the factory level so that the level of quality and reliability will be
assured for its entire life. That would ensure that the product quality and brand is
maintained at its highest level.
Thus the 2 key tests which QRA provides regularly
Accelerated Aging
Shelf Life Testing
According to the below standards for US and European markets
(a) ASTM F 1980 - 21
Standard Guide for Accelerated Ageing of Sterile Barrier Systems for Medical
Devices.
(b) ISO 11607
Stability Testing (accelerated ageing and real time ageing)

Additionally, QRA also provides the following test services and the standards complied are shown below :
- Transport Simulation
- Vibration
- Impact Shock
- Drop
- Compression
- Stacking
(a) ASTM D 4169
Performance testing for Shipping Containers and Systems
(b) ISO 11607
Package Strength and Integrity Test
Medical Device Performance and Safety Testing
Our performance and safety testing is designed to accommodate your needs for both healthcare products and laboratory equipment. Our global facilities allow us to provide efficient and custom solutions tailored to your product or facility’s requirements, and experts understand the changing market and how to best serve your business’ specific needs.
We can help you with :
- Standards testing to help you bring compliant products to market quickly
- Customized on-site support and training
- Convenient, localized testing in global facilities
- IQ OQ and PQ submissions for your in house devices
Packaging Testing
Validation of packaging procedures, such as the forming and sealing of sterile barrier systems, as per ISO 11607-2 is one of our services. Sterilized and final-packed test devices are subjected to both thermal and regular ageing, as well as transport simulation, as part of a combined stability and packaging validation study, according to ISTA or ASTM standards.
The specific qualities of medical equipment and their packaging systems must remain stable during their shelf life, according to ISO 11607-1. Following that, products can be put through a series of testing to determine the packaging system's performance, aseptic presentation, and microbiological barrier qualities, as well as the performance of the enclosed medical device, functionality, and biocompatibility.

The following test systems may be applicable for the packaging system:
- Dye test for seal integrity
- Peel test, burst test and bubble test
- Air permeability test and test for microbiological tightness
- Qualification of packaging materials
- Validation of the forming, sealing, and assembly processes of packaging for terminally sterilized medical devices
- Validation of final packaging systems for sterile medical devices
- Evaluation of the shelf life of the sterile packed medical device
- Stressed medical devices are subjected to specific performance tests as well as to a biological evaluation test strategy
